InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the “mRNA 2.0 Market”-, By Technology (Self-Amplifying mRNA (saRNA), Optimized Non-Amplifying mRNA, Circular RNA (circRNA), Programmable/Logic-Gated mRNA), By Application (Infectious Disease Vaccines, Oncology, Rare & Genetic Diseases, Autoimmune & Inflammatory Diseases, Cardiovascular & Metabolic), By End-User (Hospitals & Clinics, Research Institutions, Biopharmaceutical Companies), and Global Forecasts, 2025-2034 And Segment Revenue and Forecast To 2034.”
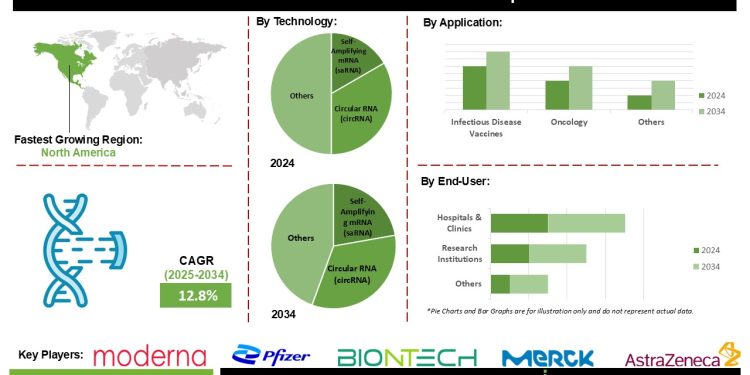
mRNA 2.0 market Size is predicted to grow at a 12.8% CAGR during the forecast period for 2025-2034.
Get Free Access to Demo Report, Excel Pivot and ToC: https://www.insightaceanalytic.com/request-sample/3111
mRNA 2.0 represents an advanced generation of messenger RNA technologies designed to overcome the limitations of earlier platforms while expanding their application beyond infectious disease vaccines. Building on the proven success of COVID-19 mRNA vaccines, this next-generation approach prioritizes improved molecular stability and enhanced delivery mechanisms. Recognizing the inherent instability of mRNA, these platforms incorporate structurally optimized RNA constructs alongside sophisticated delivery systems, including advanced lipid nanoparticles (LNPs), polymer-based carriers, and biologically derived exosomes. These delivery technologies protect RNA from enzymatic degradation, facilitate efficient cellular uptake, and reduce reliance on ultra-cold storage conditions, thereby improving global distribution and accessibility.
A key objective of mRNA 2.0 is the achievement of targeted delivery and precise protein expression within specific tissues or cell types, which is essential for applications in areas such as precision oncology, gene therapy, and other advanced therapeutic fields. As a result, mRNA 2.0 platforms are increasingly viewed as programmable tools capable of enabling controlled RNA expression with tissue-specific functionality.
Circular RNAs (circRNAs), characterized by their covalently closed single-stranded structure, are emerging as a significant component of the mRNA 2.0 landscape. Their circular configuration enhances molecular stability and supports sustained protein expression, making them particularly well suited for applications requiring prolonged biological activity, such as cancer immunotherapy and next-generation vaccines. In contrast to linear mRNA, circRNAs exhibit superior thermostability, maintaining stability at 4°C or even at ambient temperatures over extended periods. This property reduces the dependence on stringent cold chain logistics and improves feasibility in resource-constrained settings.
CircRNAs are commonly delivered through advanced lipid nanoparticle systems, including mannose-functionalized formulations that enable targeted delivery to lymphatic tissues and promote robust immune responses. In addition, lyophilized circRNA-LNP formulations retain stability after reconstitution, supporting practical use across a wide range of clinical environments.
As the mRNA therapeutics sector evolves beyond pandemic-related applications into broader therapeutic areas—such as oncology, rare genetic disorders, autoimmune diseases, cardiovascular conditions, and protein replacement therapies—circRNAs, in combination with next-generation delivery technologies like Selective Organ Targeting (SORT) LNPs, polymeric nanoparticles, and exosome-based systems, are playing a pivotal role in driving this transition. Their ability to extend protein expression, enhance stability, and reduce dosing frequency positions circRNAs as a foundational element in the advancement of RNA-based therapeutics.
Read Comprehensive Report Overview: https://www.insightaceanalytic.com/report/mrna-20-market-/3111
List of Prominent Players in the mRNA 2.0 Market:
- Moderna
- BioNTech
- Pfizer
- CureVac
- Arcturus Therapeutics
- GSK
- AstraZeneca
- Merck
- Orna Therapeutics
- Strand Therapeutics
- ReCode Therapeutics
- Abogen Biosciences
- Nutcracker Therapeutics
- Gritstone Bio
- Vertex Pharmaceuticals
- Ethris
- Ziphius Vaccines
- Replicate Bioscience
- Laronde (Senda Biosciences)
- CircBio
- Anima Biotech
- HDT Bio
- VaxEquity
- Voltron Therapeutics
- Chimeron Bio
- MiNA Therapeutics
- Acuitas Therapeutics
- Evox Therapeutics
- DSP
- eTheRNA
- Kernal Biologics
Market Dynamics
Drivers:
Advancements in RNA-based technologies, including self-amplifying RNA (saRNA), trans-amplifying RNA (taRNA), optimized non-replicating mRNA, and circular RNA (circRNA), are significantly expanding the therapeutic potential of mRNA-based approaches. These innovations enhance molecular stability, improve translational efficiency, reduce dosing requirements, and prolong protein expression, thereby broadening the applicability of mRNA therapeutics across areas such as chronic diseases, oncology, and genetic disorders.
At the same time, the development of advanced delivery systems—including Selective Organ Targeting (SORT) lipid nanoparticles, polymer-based carriers, and exosome-derived vectors—enables highly targeted delivery to specific tissues and cell types. This capability is particularly valuable for complex therapeutic indications such as cancer, neurodegenerative disorders, and metabolic diseases. Strong financial backing from government initiatives, venture capital, and strategic collaborations with pharmaceutical companies is further accelerating innovation, development, and commercialization within this space.
Challenges:
Despite significant progress, the large-scale manufacturing of mRNA therapeutics continues to face economic and technical challenges. Production processes rely on specialized and high-cost raw materials, including custom enzymes and chemically modified nucleotides, which contribute to elevated manufacturing expenses. Additionally, stringent purification requirements are necessary to eliminate contaminants such as double-stranded RNA, which can trigger unwanted immune responses. These factors introduce complexities in scaling production and may limit widespread adoption, particularly in regions with limited access to advanced biomanufacturing capabilities.
Regional Trends:
North America, led by the United States, holds a dominant position in the mRNA 2.0 market, supported by a strong scientific ecosystem, well-established regulatory frameworks, extensive research and development infrastructure, and continuous investment from both public and private sectors. These advantages position the region as a leading center for innovation and commercialization of RNA-based therapeutics.
The Asia-Pacific region is projected to witness the highest growth rate, driven by national-level initiatives aimed at developing domestic mRNA research and manufacturing capabilities. Key countries such as China, India, Japan, and South Korea are making substantial investments in infrastructure development, workforce training, and clinical research programs to enhance their global competitiveness. The rapid increase in early- and mid-stage clinical trials, along with the expansion of regional Contract Research Organizations (CROs) and Contract Development and Manufacturing Organizations (CDMOs), underscores the region’s commitment to meeting the rising demand from both domestic and international biopharmaceutical markets.
Add our site to Google Preferred Sources for quality content: https://google.com/preferences/source?q=insightaceanalytic.com
Recent Developments:
- In Apr 2025, Arcturus Therapeutics Holdings Inc. declared that the U.S. Food and Drug Administration (FDA) has awarded Fast Track Designation to ARCT-2304, a self-amplifying mRNA (sa-mRNA) vaccine candidate intended for active immunization to prevent disease caused by influenza A H5N1 subtype contained in the vaccine. The study was centered on the development of infectious disease vaccines and opportunities within liver and respiratory rare diseases.
- In Aug 2024, Abogen Biosciences, revealed a paper in Nucleic Acids Research titled “Efficient circularization of protein-encoding RNAs via a novel cis-splicing system.” The production of circular RNA (“circRNA”) with greatly increased protein expression, decreased innate immune activation, and flexible splice site design that offers significant market potential is made possible by Abogen’s highly effective RNA circularization cis-splicing system (referred to as the “Cis-System”), which was disclosed in the article. The biopharmaceutical sector has shown a great deal of interest in circular RNA technology, and a number of circular RNA start-ups have garnered more than $1 billion in funding.
Segmentation of mRNA 2.0 Market.
Global mRNA 2.0 Market – By Technology
- Self-Amplifying mRNA (saRNA)
- Optimized Non-Amplifying mRNA
- Circular RNA (circRNA)
- Programmable/Logic-Gated mRNA
Global mRNA 2.0 Market – Infectious Disease Vaccines
- Oncology
- Rare & Genetic Diseases
- Autoimmune & Inflammatory Diseases
- Cardiovascular & Metabolic
Global mRNA 2.0 Market – By End-User
- Hospitals & Clinics
- Research Institutions
- Biopharmaceutical Companies
Global mRNA 2.0 Market – By Region
North America-
- The US
- Canada
Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
Asia-Pacific-
- China
- Japan
- India
- South Korea
- Southeast Asia
- Rest of Asia Pacific
Latin America-
- Brazil
- Mexico
- Rest of Latin America
Middle East & Africa-
- GCC Countries
- South Africa
- Rest of the Middle East and Africa
Customize this Study according to your Requirements @ https://www.insightaceanalytic.com/customisation/3111
About Us:
InsightAce Analytic is a market research and consulting firm that enables clients to make strategic decisions. Our qualitative and quantitative market intelligence solutions inform the need for market and competitive intelligence to expand businesses. We help clients gain a competitive advantage by identifying untapped markets, exploring new and competing technologies, segmenting potential markets and repositioning products. expertise is in providing syndicated and custom market intelligence reports with an in-depth analysis with key market insights in a timely and cost-effective manner.
Contact us:
InsightAce Analytic Pvt. Ltd.
Visit: https://www.insightaceanalytic.com/
Tel : +1 607 400-7072
Asia: +91 79 72967118
info@insightaceanalytic.com