InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the “Global Human Vaccine Adjuvants Market Size, Share & Trends Analysis Report By Type (Pathogen, Combination, Particulate, Adjuvant emulsion, and Others), Application (Cancer, Infectious diseases, and Others), Route of Administration (Oral, Intramuscular, Intranasal, Intradermal, and Others),-Market Outlook And Industry Analysis 2035″
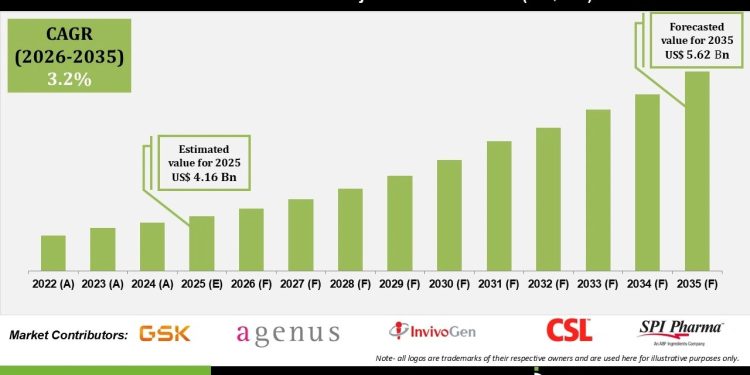
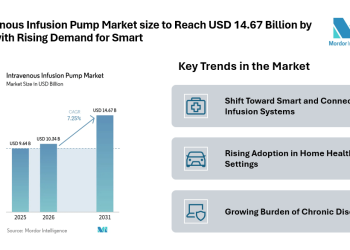
Human Vaccine Adjuvants Market Size is valued at USD 4.16 Bn in 2025 and is predicted to reach USD 5.62 Bn by the year 2035 at a 3.2% CAGR during the forecast period for 2026 to 2035.
Get Free Access to Demo Report, Excel Pivot and ToC: https://www.insightaceanalytic.com/request-sample/3441
The development of highly effective vaccines relies significantly on adjuvants, which are agents used to enhance the immune response to an antigen. These substances play a vital role in improving vaccine performance, particularly in populations where immune responses may be weaker or compromised. The growth of the Human Vaccine Adjuvants Market is being driven by the rising prevalence of infectious diseases, expanding immunization initiatives, and the continued introduction of innovative vaccines.
The market is further supported by ongoing efforts to develop vaccines for emerging infectious threats and potential pandemics. In addition, increasing global demand for safe, reliable, and effective immunization solutions is expected to contribute positively to market expansion. Growing financial investments from governments, healthcare institutions, and private organizations toward vaccine research and development are also strengthening market growth prospects.
However, the market continues to face notable challenges, particularly stringent regulatory requirements and safety concerns associated with extensive clinical evaluation. The need to demonstrate adjuvant efficacy, safety profiles, and potential health risks through comprehensive testing remains a key barrier to product development and commercialization.
Read Comprehensive Report Overview: https://www.insightaceanalytic.com/report/human-vaccine-adjuvants-market/3441
List of Prominent Players in the Human Vaccine Adjuvants Market:
- Novavax, Inc.
- GlaxoSmithKline plc.
- SPI Pharma
- Agenus, Inc.
- CSL Limited
- InvivoGen
- Brenntag Biosector
- Adjuvance Technologies, Inc.
Market Dynamics
Drivers
The Human Vaccine Adjuvants Market is experiencing significant growth, supported by expanding government immunization programs, the increasing prevalence of infectious diseases, and continuous advancements in vaccine enhancement technologies. For example, during the 2023–2024 influenza season, U.S. public health laboratories analyzed 129,638 specimens, of which 39,885 tested positive for influenza, according to the Centers for Disease Control and Prevention (CDC). This highlights the ongoing demand for more effective vaccines and advanced adjuvant systems.
In addition, regulatory authorities such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) have introduced accelerated approval pathways for vaccine innovations, supporting the commercialization of novel adjuvants and strengthening market expansion. Rising investments from both public and private sectors in vaccine research and development are further contributing to growth. Collectively, these factors, along with an increasing focus on vaccine safety and efficacy, are reshaping the global vaccine adjuvants industry.
Challenges
Despite positive market momentum, several factors continue to constrain growth. Safety concerns, including toxicity risks and adverse reactions associated with certain adjuvants, remain major challenges for manufacturers and regulatory bodies. The high cost of adjuvant research, clinical validation, and regulatory compliance also places pressure on product development timelines and commercialization efforts.
In some regions, limited public awareness regarding vaccination benefits, inadequate healthcare infrastructure, and restricted access to immunization services may further hinder market penetration. Additionally, comparatively lower research activity in certain developing markets may slow innovation and adoption, thereby limiting overall market growth.
Regional Trends
In 2025, North America accounted for the largest share of the Human Vaccine Adjuvants Market. The region’s leadership is supported by advanced healthcare infrastructure, substantial government funding for vaccine development, and strong adoption of innovative vaccine technologies. The United States has played a central role in this growth due to its established biotechnology and pharmaceutical research ecosystem. Major pharmaceutical companies and research institutions across the region are actively engaged in the development and production of adjuvanted vaccines.
North America’s market position is further reinforced by robust regulatory systems, high healthcare expenditure, and continued investment in vaccine programs targeting diseases such as COVID-19 and influenza. In addition, growing demand for cancer immunotherapy vaccines and personalized medicine solutions is creating new growth opportunities across the region.
Asia Pacific is projected to register the fastest growth during the forecast period. Expansion is being driven by the healthcare needs of large populations, rising government expenditure on vaccine development, and increasing adoption of immunization programs. Countries such as China, India, and Japan are major contributors to regional growth, supported by the rising burden of infectious diseases and stronger vaccination initiatives.
Furthermore, the region’s expanding biotechnology sector is enabling the development and manufacturing of advanced adjuvants, particularly for emerging diseases. Increasing public-private partnerships in vaccine research and development, together with rising vaccine demand across both developed and emerging economies, are expected to accelerate market growth throughout Asia Pacific.
Add our site to Google Preferred Sources for quality content: https://google.com/preferences/source?q=insightaceanalytic.com
Recent Developments:
- October 2024: GSK plc released new data from the AReSVi-006 phase III trial, which assessed the effectiveness of a single dose of Arexvy, a recombinant adjuvanted respiratory syncytial virus vaccine, against lower respiratory tract disease (LRTD) caused by RSV in adults 60 years of age and older, including those at increased risk over three complete RSV seasons (NCT04886596).
- May 2024: The first doses of the R21/Matrix-M malaria vaccine were sent to African nations, starting with the Central African Republic, by the Serum Institute of India in partnership with Novavax and the University of Oxford. By utilizing the saponin-based Matrix-M adjuvant for increased immunogenicity, this is a significant step toward entering endemic areas.
- March 2023: The acquisition of CENTAURI Technologies was announced by Aurorium. The addition of CENTAURI’s sophisticated manufacturing capabilities and complementing specialized material product portfolio broadens Aurorium’s offerings and increases its presence in high-growth end markets.
Segmentation of Human Vaccine Adjuvants Market-
By Type-
- Pathogen
- Combination
- Particulate
- Adjuvant emulsion
- Others
By Application-
- Cancer
- Infectious diseases
- Others
By Route of Administration-
- Oral
- Intramuscular
- Intranasal
- Intradermal
- Others
By Region-
North America-
- The US
- Canada
Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
Middle East & Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
Customize this Study according to your Requirements @ https://www.insightaceanalytic.com/customization/3441
About Us:
InsightAce Analytic is a market research and consulting firm that enables clients to make strategic decisions. Our qualitative and quantitative market intelligence solutions inform the need for market and competitive intelligence to expand businesses. We help clients gain competitive advantage by identifying untapped markets, exploring new and competing technologies, segmenting potential markets and repositioning products. Our expertise is in providing syndicated and custom market intelligence reports with an in-depth analysis with key market insights in a timely and cost-effective manner.
Contact us:
InsightAce Analytic Pvt. Ltd.
Visit: https://www.insightaceanalytic.com/
Tel : +1 607 400-7072
Asia: +91 79 72967118
info@insightaceanalytic.com